 Daily Newsletter
Daily Newsletter
September 24, 2014
Redox and Coupled Reactions
Energy harvesting will be our topic next week, so I wanted to spend a moment and talk about energy in biological systems. Energy is a word that is often thrown around in various disciplines, and most of us carry misconceptions about the world from colloquial (common) use of the word. Scientists define energy as the capacity to do work. This definition helps to simplify a complex issue, but it starts to get confused when we apply it to the complex sets of reactions that we see in living systems.
For example, as we saw yesterday, the work in phosphorylation occurs in creating the covalent bond between a phosphate group and a substrate, not the action of the substrate. With ATP, the emphasis was shifted from seeing ATP as a battery that powered reactions. Instead, your focus was drawn to the Phosphate group, and the electrostatic effect it would have when added to a molecule or protein.
Next week we will discuss Energy Harvesting. Like with ATP, I want you to focus your attention on a specific form of energy, instead of holding a nebulous concept. Today the focus will be on reducing potential. Central metabolism describes the oxidation of glucose, so what are we harvesting? Reducing potential. So what is reducing potential?
A simple definition is reducing potential describes the capacity of a compound to donate electrons. Chemistry has a strict definition involving measurements with electrodes, but for our purpose, the concept of donating electrons is what is important.
Remember the characteristics of life. You must maintain homeostasis, and this means repair. You have to build nucleic acids, lipids, carbohydrates and proteins. These biosynthetic pathways often require you to reduce substrates. To stay alive, you need a constant supply of electrons for reduction; you need reducing potential. If you don't get these high energy electrons for reduction, you die. We will also find that this reducing potential is needed for us to make ATP.
Redox reactions are vital to our survival. Redox reactions are coupled Oxidation and Reduction reactions. One compound is oxidized as the next is reduced.
For example, as we saw yesterday, the work in phosphorylation occurs in creating the covalent bond between a phosphate group and a substrate, not the action of the substrate. With ATP, the emphasis was shifted from seeing ATP as a battery that powered reactions. Instead, your focus was drawn to the Phosphate group, and the electrostatic effect it would have when added to a molecule or protein.
Next week we will discuss Energy Harvesting. Like with ATP, I want you to focus your attention on a specific form of energy, instead of holding a nebulous concept. Today the focus will be on reducing potential. Central metabolism describes the oxidation of glucose, so what are we harvesting? Reducing potential. So what is reducing potential?
A simple definition is reducing potential describes the capacity of a compound to donate electrons. Chemistry has a strict definition involving measurements with electrodes, but for our purpose, the concept of donating electrons is what is important.
Remember the characteristics of life. You must maintain homeostasis, and this means repair. You have to build nucleic acids, lipids, carbohydrates and proteins. These biosynthetic pathways often require you to reduce substrates. To stay alive, you need a constant supply of electrons for reduction; you need reducing potential. If you don't get these high energy electrons for reduction, you die. We will also find that this reducing potential is needed for us to make ATP.
Redox reactions are vital to our survival. Redox reactions are coupled Oxidation and Reduction reactions. One compound is oxidized as the next is reduced.
Remember, the molecules undergoing redox have to be close/touching. But in relative size, a cell is huge compared to a simple molecule. We may harvest electrons (oxidation) in one part of the cell, but use the harvested reducing potential in another part of the cell (reduction). Remember, you don't have free electrons; you can't throw electrons across the cytoplasm. So, how do we couple reactions that may be separated spatially? We use carriers!
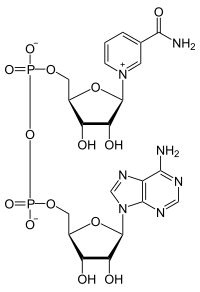
Electron carriers, like nicotinamide adenine dinucleotide (NAD+), accept electrons at the site of oxidation, and then donate electrons at the site of reduction. NAD+ is readily oxidized and reduced during metabolic reactions, and there is only a negligible loss of energy from the electrons carried (can we ever have NO loss of energy? why or why not?).
NAD+ is also classified as a coenzyme, meaning it must work with an enzyme to accept or donate electrons. NAD+ can not randomly go to a molecule and oxidize or reduce it; its action is regulated by enzymes. NAD+ then must bind to an enzyme that catalyzes an Oxidation, and NADH must bind to an enzyme that catalyzes a Reduction.

Specifically, we couple the reactions. NAD+ has a place to bind into the enzyme, many times next to the substrate. The NAD+ can then capture the eletron pair that is released from the substrate. Additionally, one of the hydrogens will bind to the electron carrier. When we move to the next reaction, NADH will bind with an enzyme, again normally next to the substrate in question. The NADH can then donate the electrons (and hydrogens to the substrate. Enzymes thus help to couple these reactions. NAD+ will not just pick up an electron from any source, and NADH will not just donate electrons to any source. It must be mediated by enzymes.

Electron carriers, like nicotinamide adenine dinucleotide (NAD+), accept electrons at the site of oxidation, and then donate electrons at the site of reduction. NAD+ is readily oxidized and reduced during metabolic reactions, and there is only a negligible loss of energy from the electrons carried (can we ever have NO loss of energy? why or why not?).
NAD+ is also classified as a coenzyme, meaning it must work with an enzyme to accept or donate electrons. NAD+ can not randomly go to a molecule and oxidize or reduce it; its action is regulated by enzymes. NAD+ then must bind to an enzyme that catalyzes an Oxidation, and NADH must bind to an enzyme that catalyzes a Reduction.

Specifically, we couple the reactions. NAD+ has a place to bind into the enzyme, many times next to the substrate. The NAD+ can then capture the eletron pair that is released from the substrate. Additionally, one of the hydrogens will bind to the electron carrier. When we move to the next reaction, NADH will bind with an enzyme, again normally next to the substrate in question. The NADH can then donate the electrons (and hydrogens to the substrate. Enzymes thus help to couple these reactions. NAD+ will not just pick up an electron from any source, and NADH will not just donate electrons to any source. It must be mediated by enzymes.
Daily Challenge
This week you must complete all four forums to be eligible for the weekly summary and summative quiz.
Completion requires that you start a discussion, that your discussion is a minimum of 150 words on topics and at a collegiate level of writing, and that you reply to 3 of your fellow students.
Forum Closes: Septembe 25, 2014 at 11:55pm
Completion requires that you start a discussion, that your discussion is a minimum of 150 words on topics and at a collegiate level of writing, and that you reply to 3 of your fellow students.
Forum Closes: Septembe 25, 2014 at 11:55pm
Action of nicotinamide adenine dinucleotide (NAD)
In the citric acid cycle is the following reaction:
In the citric acid cycle is the following reaction:
In this reaction, malate is oxidized. How do you know? You know because NAD is reduced to NADH. Below is a ribbon model of the protein malate dehydrogenase. Within the protein, you will see two molecules of NAD represented as balls. NAD binds to the enzymes active site first, and then malate binds. Within the active site are both + and - amino acids.
Your task today, using the enzyme malate dehydrogenase, explain how enzymes work and explain how reducing potential is harvested from organic compounds.
Your task today, using the enzyme malate dehydrogenase, explain how enzymes work and explain how reducing potential is harvested from organic compounds.


A GREAT SPELL CASTER (DR. EMU) THAT HELP ME BRING BACK MY EX GIRLFRIEND.
ReplyDeleteAm so happy to testify about a great spell caster that helped me when all hope was lost for me to unite with my ex-girlfriend that I love so much. I had a girlfriend that love me so much but something terrible happen to our relationship one afternoon when her friend that was always trying to get to me was trying to force me to make love to her just because she was been jealous of her friend that i was dating and on the scene my girlfriend just walk in and she thought we had something special doing together, i tried to explain things to her that her friend always do this whenever she is not with me and i always refuse her but i never told her because i did not want the both of them to be enemies to each other but she never believed me. She broke up with me and I tried times without numbers to make her believe me but she never believed me until one day i heard about the DR. EMU and I emailed him and he replied to me so kindly and helped me get back my lovely relationship that was already gone for two months.
Email him at: Emutemple@gmail.com
Call or Whats-app him: +2347012841542